|
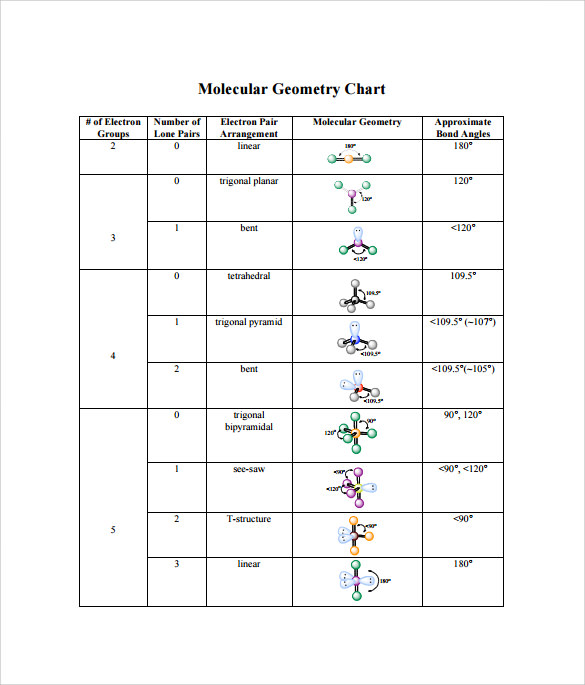
The oxygen has two bonding electron pairs (single bond to each H) and two non-bonding pairs giving water a #AX_2E_2# conformation and a bent shape. #H_2O# we need to consider the central atom of water which is oxygen. We can use the following notations when examining a Lewis structure of a molecule.Į = non-bonding electron pairs of the central atom This theory basically says that bonding and non-bonding electron pairs of the central atom in a molecule will repel (push away from) each other in three dimensional space and this gives the molecules their shape. VESPR stands for valence shell electron pair repulsion. It applies a theory called VESPR for short. Molecular geometry is a way of describing the shapes of molecules. 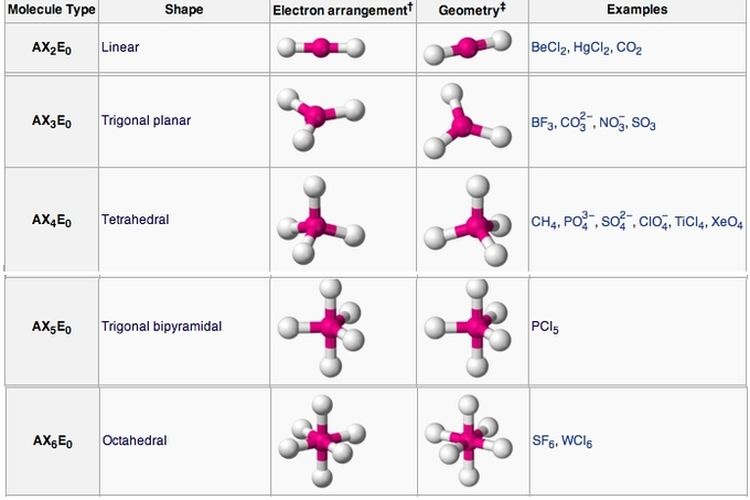
Similar logic applies to all the shapes, you just have to remember which "spoke" will be taken up by an electron pair. Once there are any electron pairs, one spoke of the original shape gets "eaten up": for example, a #AX_4E_2# is an octahedron shape, but the two "spokes" are taken up by electron pairs, so you're left with just the square-a square planar shape. #6#: octahedron (a flat square with two "spokes") #5#: trigonal bipyramid (a trigonal planar shape with two "spokes") #3#: trigonal plane (a flat equilateral-triangle-looking shape) As it has a VSEPR shape #AX_5E_0# it is a trigonal bipyramid.Įach steric number has a same "basic shape": Its steric number is #5# due to the #5# bonded atoms to the central #S# atom plus #0# lone electron pairs. Thus, it is in the form #AX_3E_1#, which forms a trigonal pyramidal shape. #N#, the central atom, has a steric number of #4#, calculated by the #3# atoms it's bonding with #+1# lone pair. This is the total number of electron pairs and bonds with other atoms. It differs depending on the shape of the molecule.Find the central molecules' steric numbers. A bond angle is a theoretical angle between two adjoining bonds. There are several types of hybridization: sp, sp2, sp3. If you may determine the angles between hybrid orbitals, it will help to explain to you a molecule shape, because these angles are nearly equal to the angles between bonds. This column contains the general molecular shape and other parameters, such as lengths and angles of bonds that demonstrate the location of each atom. To calculate this number, you should summarize all the E in the generic formula. This column represents the amount of lone pairs, which is a place with non-bonding electrons. To calculate this number, you should summarize all the X in the generic formula. This column contains the amount of connected atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
